ALDEHYDES AND KETONES
Aldehydes and Ketones are characterised by the presence of Carbonyl group >C = O in their molecules. Aldehydes contain group 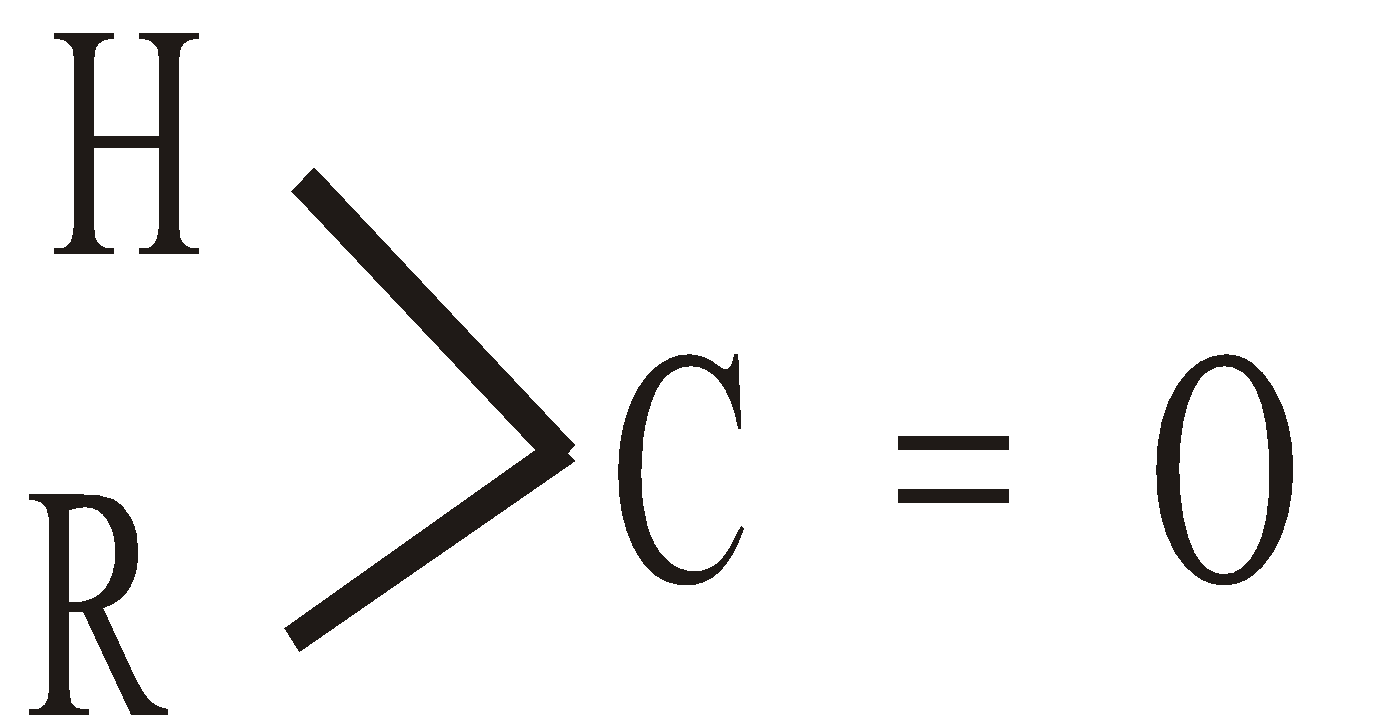 and ketones the
and ketones the 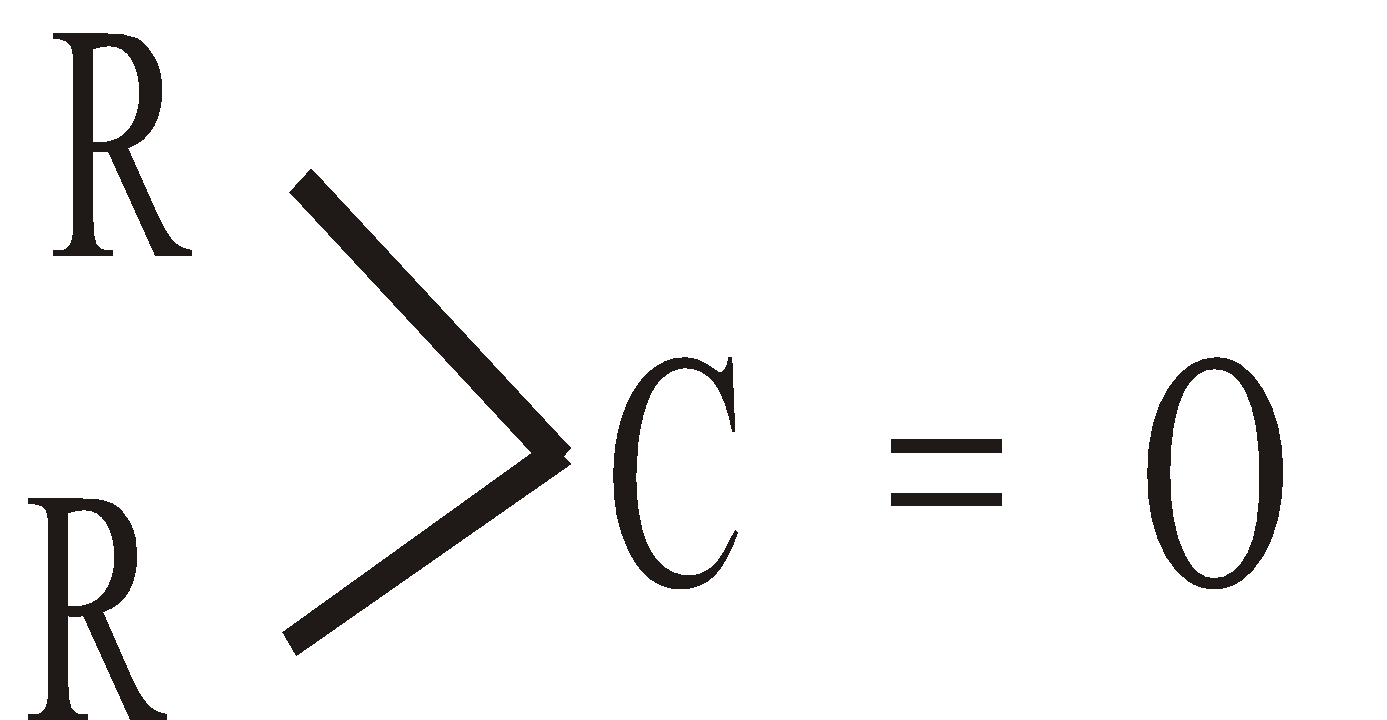 . If the groups attached to carbonyl carbon are the same, the ketone is symmetrical and if they are different the ketone is unsymmetrical.
. If the groups attached to carbonyl carbon are the same, the ketone is symmetrical and if they are different the ketone is unsymmetrical.
NATURE OF CARBONYL GROUP
The carbon and oxygen of the carbonyl group are sp2 hybridised and the carbonyl double bond is made of one s bond and one p bond.
The electronegativity of oxygen is much higher than that of carbon, the p electron cloud is displaced towards the oxygen. Therefore the C–O bond is polar in nature and carbonyl compounds possess dipole moment (2.3 to 2.8 D)
NOMENCLATURE OF ALDEHYDES
There are two systems
- Common system : The suffix “-ic acid” is replaced by the suffix “-aldehyde”
- IUPAC system : The suffix “-e” of alkane is replaced by the suffix “-al”.
Compound Common name IUPAC name
HCHO Formaldehyde Methanal
CH3CHO Acetaldehyde Ethanal
NOMENCLATURE OF KETONES
- Common system : Symmetrical ketones are named as dialkyl ketone and name of unsymmetrical ketone is obtained by naming the alkyl groups alphabetically and adding the third word ketone.
- IUPAC system : The suffix “-e” of corresponding alkane is replaced by “-one”
Compound Common name IUPAC name
H3C.COCH3 dimethyl ketone (acetone) Propanone
H3C.COC2H5 ethyl methyl ketone butanone
In higher ketones the numbering of C-atoms is must to show the position of carbonyl group. eg. :
3-hexanone
2-methyl-4-heptanone
ISOMERISM IN ALDEHYDES
Aldehydes exhibit two types of isomerism
Chain isomerism
Functional isomerism
ISOMERISM IN KETONES
They exhibit three types of isomerism
Chain isomerism
Functional isomerism
Position isomerism
GENERAL METHODS OF PREPARATION OF ALDEHYDES
- Controlled oxidation of primary alcohols
Oxidising agents K2Cr2O7/H2SO4 or KMnO4/H2SO4
PCC is pyridinium chlorochromate known as Collin’s reagent and is specific for oxidation of 1º alcohol to aldehyde.
- Dehydrogenation of primary alcohol
- By Rosenmund reduction from acid chloride
- Hydration of Alkynes (Kucherov reaction)
- Reductive ozonolysis of alkenes
- Distillation of Calcium salt of fatty acid with calcium formate
- From acid
- Waker’s process
- Oxo process
- Stephen’s reduction of nitriles :
- Hydrolysis of Geminal halides
- From Glycols
- From Grignard’s reagent
- From acid chloride by the use of lithium t-butoxy aluminium hydride
GENERAL METHODS OF PREPARATION OF KETONES ONLY
- Oxidation of 2º alcohol
Oxidising agent K2Cr2O7/H2SO4 or KMnO4/H2SO4
- Dehydrogenation of 2º alcohols
- Hydration of alkynes
- Reductive ozonolysis of alkenes
- From Calcium salt of an acid
- From acid
- Hydrolysis of non-terminal gem.halide
- From Grignard’s reagents
- From acid chlorides
PHYSICAL PROPERTIES
- Formaldehyde is a gas and its 40% aqueous solution was known as formalin but now it is 40% HCHO, 8% CH3OH, 52% H2O. They are polar in nature and have higher values of b.p. Lower members are soluble in water.
- Lower aldehydes and ketones (C1 – C4) are soluble in water due to presence of H–bonding.
- Reactivity
It is due to + I effect of alkyl groups which decreases the +ve charge on carbonyl carbon.
Steric hindrance : The bulky alkyl group hinder the approach of nucleophile.
a-hydrogen atom is acidic in nature due to Resonance
CHEMICAL PROPERTIES
ADDITION REACTIONS
Their addition reactions are known as nucleophilic addition reactions
The followings reaction in different ways with NH3
It is used as urinary antiseptic.
NUCLEOPHILIC ADDITION REACTIONS WITH ELIMINATION OF WATER MOLECULE
The control of pH is must for these reactions. The optimum value is around 3.5.
Control of pH during formation of ammonia derivatives :
At low pH : H+ concentration is very high. The carbonyl compound and ammonia derivative, both protonated and latter cannot act as nucleophile .
At high pH : H+ concentration is too small. The protonation of carbonyl group will not occur and reaction will not occur smoothly.
Hence, optimum pH of the medium is around 3.5.
OXIDATION
Ketones are oxidised by strong oxidising agents such as Conc. HNO3, K2Cr2O7/H2SO4, KMnO4/H2SO4
OXIDATION WITH SeO2
The CH3 group adjacent to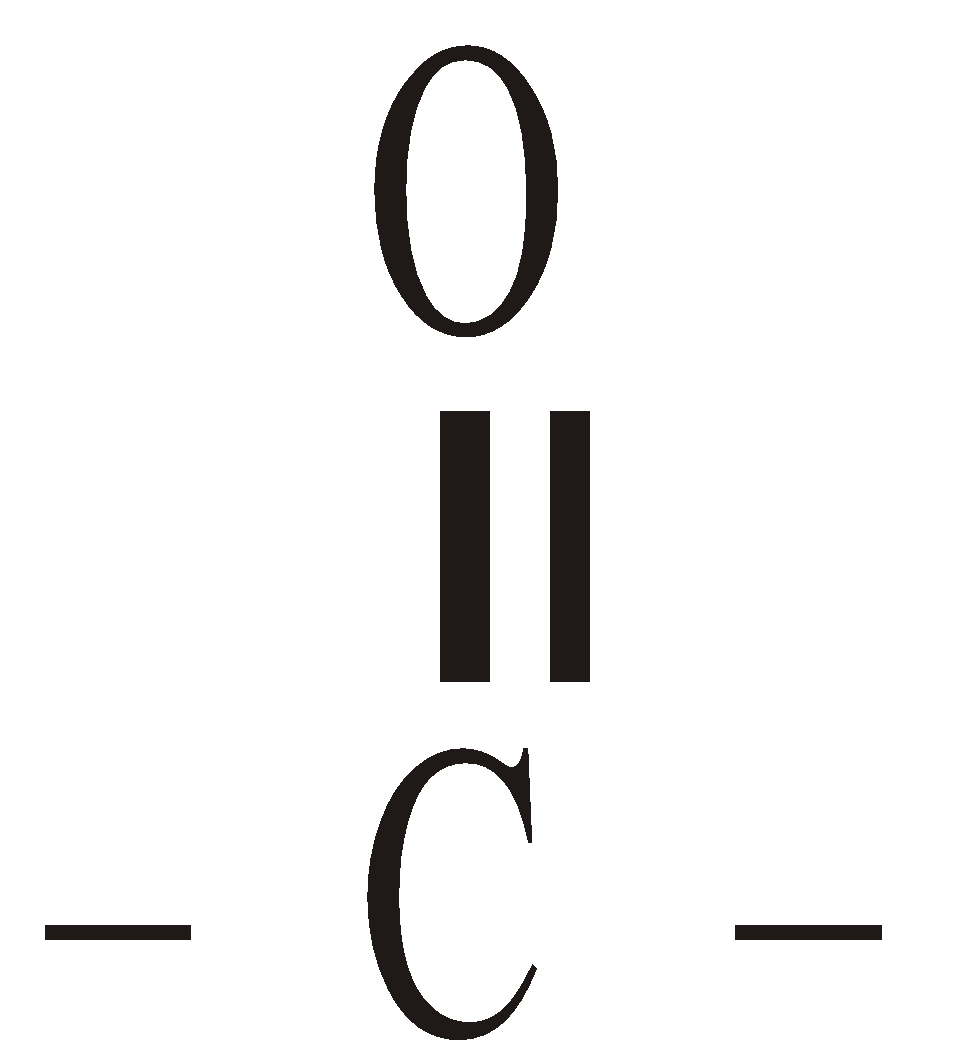 is oxidised to – CHO and >CH2 group is oxidised to
is oxidised to – CHO and >CH2 group is oxidised to
>C = O group.
The CH3 group adjacent to
>C = O group.
POPOFF’S RULE
During oxidation of unsymmetrical ketone the carbonyl group is retained by smaller alkyl group.
During oxidation of unsymmetrical ketone the carbonyl group is retained by smaller alkyl group.
Jone’s reagent : Acidified K2Cr2O7 i.e. chromic acid and sulphuric acid mixture
HALOFORM REACTION
Oxidation of acetaldehyde or methyl ketones with Sodium Hypohalite (NaOX) or (X2 + NaOH) gives haloform CHX3. The reaction is of practical value to identify these compounds by forming CHI3.
BAYER-VILLIGER OXIDATION
Oxidation of aliphatic Ketones by organic per acids, e.g. perbenzoic acid, peracetic acid or monoperthalic acid to form esters or their hydrolysed products
REDUCTION
CONDENSATION REACTIONS
ALDOL CONDENSATION
Aldehydes and Ketones having at least one a-hydrogen atom in presence of dil. alkali give b- hydroxy aldehyde or b-hydroxy ketone, which on heating gives a,b-unsaturated carbonyl compound.
CROSSED ALDOL CONDENSATION
INTRAMOLECULAR ALDOL CONDENSATION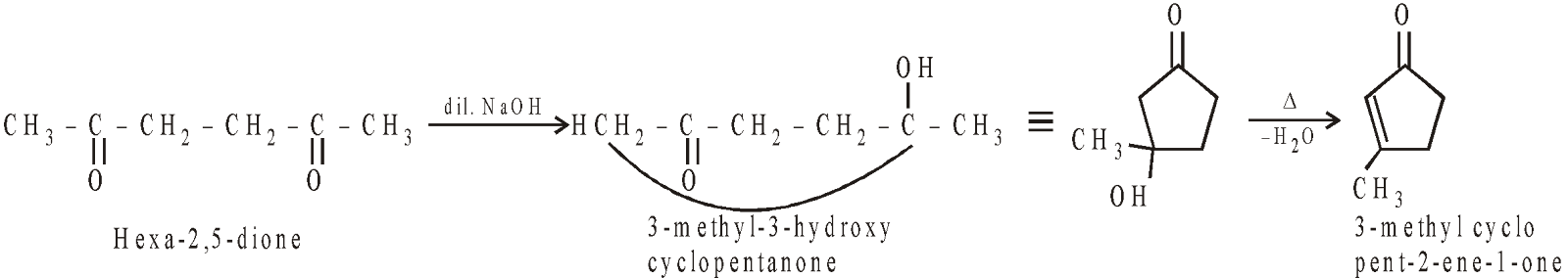
CANNIZZARO’S REACTION
Aldehydes containing no a-hydrogen atom on warming with 50% NaOH or KOH undergo disproportionation i.e. self oxidation - reduction known as cannizzaro’s reaction.
* 2-methyl propanal (CH3)2CH.CHO has a-hydrogen atom but gives Cannizzaro’s reaction.
Aldehydes containing a-H atoms on heating with conc. alkali give brown resinous mass by undergoing repeated aldol condensation.
CROSSED CANNIZZARO’S REACTION
TISHCHENKO REACTION
Aldehydes containing a-hydrogen atom with aluminium ethoxide give esters.
REFORMATSKY REACTION
It is the reaction between an a-bromo acid ester and a carbonyl compound (aldehyde or ketone) in the presence of zinc to form a b-hydroxy ester.
BECKMANN’S REARRANGEMENT
It is rearrangement of keto oxime to N-substituted acid amide in presence of Conc. H2SO4, PPA, SOCl2, PCl5 etc.
CONDENSATION PRODUCTS OF ACETONE
Above reactions are not aldol condensations.
POLYMERS OF FORMALDEHYDES
POLYMERS OF ACETALDEHYDE
BENZALDEHYDE (AROMATIC ALDEHYDES)
PREPARATION
- Etard’s oxidation
- Oxidation of toluene
- Gattermann Koch Synthesis
(Combination of CO + HCl act as formyl chloride HCOCl)
It is an electrophilic substitution reaction, the electrophile 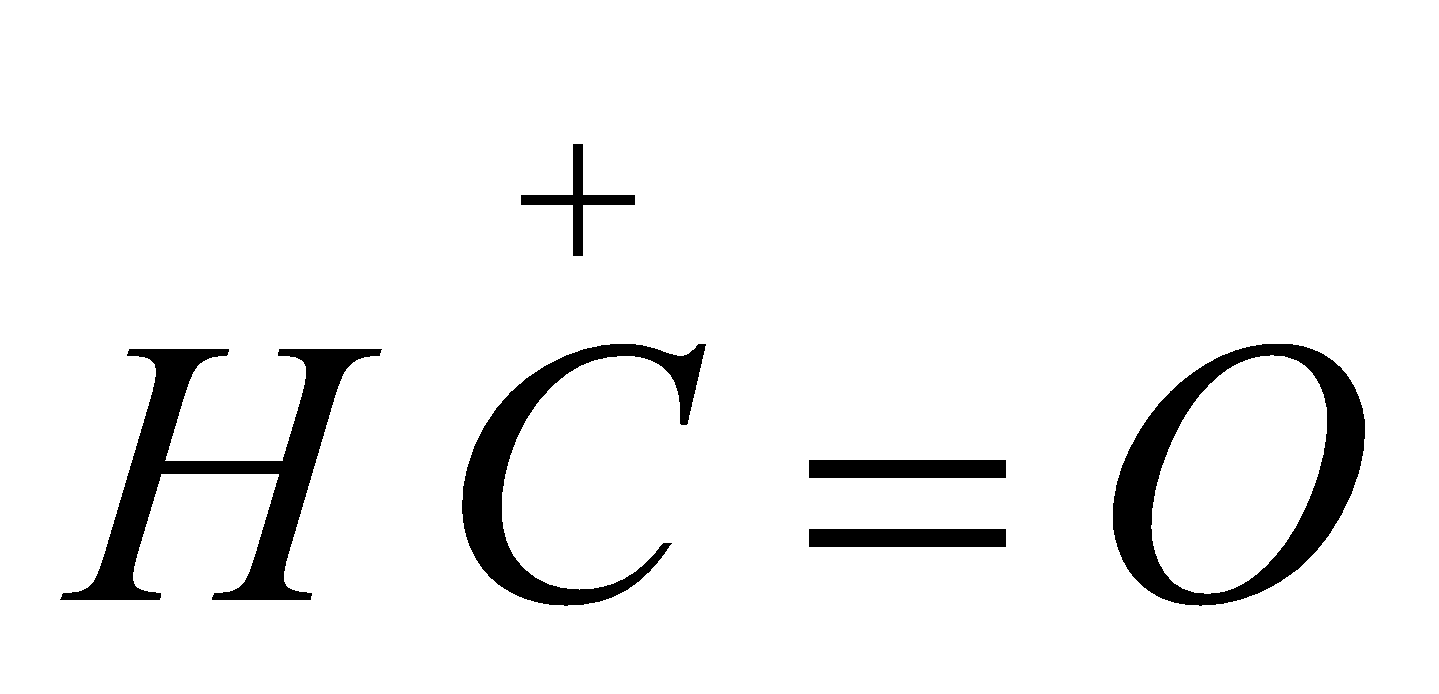 is formylium ion.
is formylium ion.
- Gattermann aldehyde synthesis
- From benzyl chloride (Lab method)
- Rosenmund reaction
- Vilsmeyer reaction
- From Grignard’s reaction
- From Benzal chloride (manufacture)
- Partial oxidation of toluene (manufacture)
PHYSICAL PROPERTIES
Colourless, highly refractive liquid b.p. 443K. It has smell of bitter almonds, slightly soluble in water. Steam volatile and poisonous.
CHEMICAL PROPERTIES
REACTION OF CHO GROUP GIVEN BY AROMATIC ALDEHYDES
REACTIONS DUE TO BENZENE NUCLEUS
CHO group is meta directing with deactivation of benzene nucleus
Benzaldehyde reduces Tollen’s reagent but not Fehling solution.
USES
- As flavouring agent
- In perfumes
- Manufacture of triphenylmethane dyes
ACETOPHENONE (AROMATIC KETONES)
PREPARATION
- By Friedel Craft’s acylation
- From benzaldehyde
- Manufacture
PHYSICAL PROPERTIES
It is colourless crystalline compound m.p. 20ºC, b.p. 202ºC.
At room temperature it is coloured liquid.
CHEMICAL PROPERTIES
- Reactions due to
. It gives almost all reactions due >C = O group.
- Reactions due to benzene nucleus : –COCH3 is meta directing in nature. Hence electrophilic substitution reactions give meta derivative.
- It give iodoform test.
- Some important reactions are :
Its vapour attack nose, throat and lungs. It is used as tear gas.
USES
It is used as hypnotic in medicine.
TEST FOR ALDEHYDE
- Tollen’s reagent (ammoniacal silver nitrate). All aldehydes give silver mirror.
- Fehling Solution (alkaline solution of Cu2+ complexed with Sodium potassium tartrate blue colour). All aldehydes give red colour except benzaldehyde.
- Benedict’s solution (Copper sulphate, sodium citrate and sodium carbonate solution). Aldehydes give reddish brown ppt.
- Schiff’s reagent (dilute solution of p-rosaniline hydrochloride decolourised with SO2 or H2SO4). Aldehydes give pink colour.
Acetone respond to this test slowly and o-hydroxy benzaldehyde does not give pink colour with schiff’s reagent
Note : Formic acid, tartaric acid, a-hydroxy ketones, glucose, fructose reduce Tollen reagent and Fehling solution.










