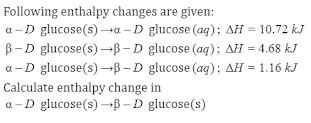IIT JEE exam which consists of JEE Main and JEE Advanced is one of the most important entrance exams for engineering aspirants. The exam is held for candidates who are aspiring to pursue a career in the field of engineering and technical studies.
Chemistry is important because everything you do is chemistry! Even your body is made of chemicals. Chemical reactions occur when you breathe, eat, or just sit there reading. All matter is made of chemicals, so the importance of chemistry is that it's the study of everything..
Chemistry is important because everything you do is chemistry! Even your body is made of chemicals. Chemical reactions occur when you breathe, eat, or just sit there reading. All matter is made of chemicals, so the importance of chemistry is that it's the study of everything..
Q1.The standard heat of formation of U3O8 is -853.5 kcal mol-1 and standard heat of the reaction, 3UO2 + O2 → U3O8 is - 76.01 kcal. The standard, heat of formation of UO2 is/are ::
Solution
-259 kcal mol-1
-259 kcal mol-1
Q3.All natural processes proceed spontaneously in a direction which:
Solution
Decreases free energy
Decreases free energy
Q4.Hess’ law is applicable for determination of enthalpy of
Solution
All of these
All of these
Q5.Which is an irreversible process?
Solution
All of the above
All of the above
Q6.Which of the following statements is/are false?
Solution
Work is not a state function, but it is a path function
Work is not a state function, but it is a path function
Q7.Work done on the gas in single stage compression is
Solution
pV = nRT
2×8 = 2×0.080×T
Wirr = -pext(V2-V1)
= -20(nRT/p2 - nRT/p1) = 144 bar-L
pV = nRT
2×8 = 2×0.080×T
Wirr = -pext(V2-V1)
= -20(nRT/p2 - nRT/p1) = 144 bar-L
Q8.The specific heat of a gas at constant volume is 0.075 cal/g. Predict the atomicity of the gas. Molar mass of gas is 40 g mol-1
Solution
CV = 0.075*40 = 3 cal
Cp-CV = 4
Cp - 3 = 2
Cp = 5 cal
γ = Cp/CV = 5/3 = 1.66
Thus, gas is monatomic
CV = 0.075*40 = 3 cal
Cp-CV = 4
Cp - 3 = 2
Cp = 5 cal
γ = Cp/CV = 5/3 = 1.66
Thus, gas is monatomic
Q9.When CaCO3 is heated to a high temperature, it undergoes decomposition into CaO and CO2 whereas it is quite stable at room temperature. The most likely explanation of it is
Solution
∆G = ∆H - T∆S, at high temperature T∆S factor dominates of ∆H and hence ∆G becomes negative and reaction occurs spontaneously
∆G = ∆H - T∆S, at high temperature T∆S factor dominates of ∆H and hence ∆G becomes negative and reaction occurs spontaneously
Q10. Given that:
A(s) ⟶ A(l) ; ∆H = x
A(l) ⟶ A(g) ; ∆H = y
The heat of sublimation of A will be
A(s) ⟶ A(l) ; ∆H = x
A(l) ⟶ A(g) ; ∆H = y
The heat of sublimation of A will be
Solution
A(s) ⟶ A(l) ; ∆H = x
A(l) ⟶ A(g) ; ∆H = y
_____________________________________________
On addition A(s) ⟶ A(g) ; ∆H = x + y
_________________________________________________
A(s) ⟶ A(l) ; ∆H = x
A(l) ⟶ A(g) ; ∆H = y
_____________________________________________
On addition A(s) ⟶ A(g) ; ∆H = x + y
_________________________________________________