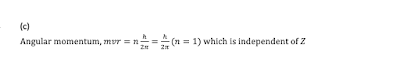JEE Advanced Physics Syllabus can be referred by the IIT aspirants to get a detailed list of all topics that are important in cracking the entrance examination. JEE Advanced syllabus for Physics has been designed in such a way that it offers very practical and application-based learning to further make it easier for students to understand every concept or topic by correlating it with day-to-day experiences. In comparison to the other two subjects, the syllabus of JEE Advanced for physics is developed in such a way so as to test the deep understanding and application of concepts.
Q1. The maximum angular speed of the electron of a hydrogen atom in a stationary orbit is
Q2. In interpreting Rutherford’s experiments on the scattering of alpha particles by thin foils, one must examine what were the known factors, and what did the experiment conclude. Which of the following are true in this context?
Q3. An electron jumps from the 4th orbit to the 2nd orbit of hydrogen atom. Given: the Rydberg’s constant =105 cm(-1). The frequency, in Hz, of the emitted radiation will be
Q5. A neutron moving with a speed v makes a head-on collision with a hydrogen atom in ground state kept at rest. The minimum kinetic energy of the neutron for which inelastic collision will take place is (assume that mass of proton is nearly equal to the mass of neutron)
Q6. An electron and a photon have same wavelength. If p is the momentum of electron and E the energy of photon, the magnitude of p/E is S.I. unit is
Q7. An electron revolving in an orbit of radius 0.5 Å in a hydrogen atom executes 1016 revolutions per second. The magnetic moment of electron due to its orbital motion will be
Q8. The frequency of emission line for any transition in positronium atom (consisting of a positron and an electron) is x times the frequency for the corresponding line in the case of H atom, where x is
Solution
(d) Rydberg’s constant determines the frequencies. We have R∝m. So, modified R for positronium atom is half of H atom. Hence, frequencies are reduced to half
(d) Rydberg’s constant determines the frequencies. We have R∝m. So, modified R for positronium atom is half of H atom. Hence, frequencies are reduced to half
Q9. If the potential difference applied across a Coolidge tube is increased, then
Solution
(c) The characteristic X-ray depends on the material used
(c) The characteristic X-ray depends on the material used
Q10. If R is the Rydberg constant for hydrogen, then the wave number of the first line in the Lyman series is